Step therapy is a process that requires physicians to prescribe more costly drugs, usually brand drugs, only after a less costly drug treatment has been tried and failed with a patient. Step therapy is just one tool in the drug utilization management (DUM) toolkit which includes drug utilization review, quantity limits, prior authorization, refill too soon, and mandatory generic enforcement programs just to name a few. Step therapy programs evoke strong emotions in both support and opposition.
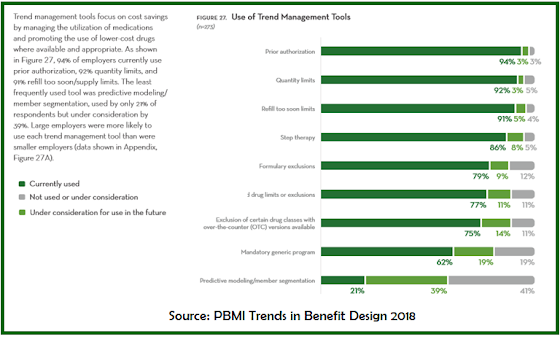 |
| Click to Enlarge |
Positions in Opposition of Step Therapy Programs
Coalition of State Rheumatology Organizations: Step therapy can lead to serious negative patient outcomes and increased costs if not carefully managed. Step therapy needs to be regulated by nationally recognized clinical practice guidelines. Physicians should have the authority to override step therapy protocols based on a patient’s medical history. Override protocols should be clear and abbreviated.
Patient Rising: Step therapy prevents patients from accessing treatments prescribed by their doctor, and instead mandates a course of treatment mandated by their insurance carrier. Patients start with older, cheaper treatments, and if they are not effective, patients “step” to another treatment. Some patients suffer terribly during this process, becoming sicker when denied prescribed treatments. For this reason, it becomes abundantly clear why patients across the country have another description for step therapy: “fail first.” When a patient has to fail first on a drug before being allowed to take the medication originally prescribed, the patient, physician and public health suffers.
National Psoriasis Foundation: Because of the rise in costs of conventional medical treatments, many patients nowadays are beguiled by their health insurance companies into undergoing considerably less expensive options such as step therapy. This “wealth before health” kind of thinking is actually futile and unwise, at best, since step therapy is never a good option in treating life-threatening diseases.
According to a new study, led by researchers at Tufts University, tracked the application of step therapy protocols across 17 payers. Across those insurers, 38.9% of coverage policies deployed some kind of step therapy protocol. On average, insurers required 1.5 steps in their protocols, with 66.6% of policies requiring a single step. Of the remaining policies, 22.7% required two steps, 7.6% were three steps and 3.1% included four or more steps, according to the study.
Here’s the part few opponents talk about
Before I owned a mail-order pharmacy and
fiduciary-model PBM, I was a pharmaceutical sales representative. Yes, whatever you’ve heard about drug sales reps it is likely true. It was a good gig with a great company. Eli Lilly and Company had a big sales force and effective marketing machine behind it. It took me 1.5 years but I took a territory ranked something like 600 out of 612 to #12 in the country.
I did it by asking doctors to prescribe a new brand oral antidiabetic, Actos, as a first line drug therapy or in combination with metformin. It wasn’t any more complicated than that.
Looking back, there were thousands of prescriptions written for an expensive brand drug when the “old” tried and true generic metformin would have been as efficacious at 1/100 the cost. Multiply that by the tens of thousands of other drug sales reps across the country doing exactly the same thing. I’ll dig deeper into direct-to-consumer advertising as a key sales driver in a separate post. But don’t kid yourself. A lot of people see a TV ad then go into the doctor’s office and say, “I want to try that.” Simply put, pharmacy benefit managers deploy step therapy as a tool to manage drug costs.
Step therapy programs are not created equally. Some PBMs run better, more efficient step therapy programs than others. That said, no PBM wants to put patients in harms way. Opponents of step therapy programs aren’t cutting the checks which cover the high costs of biologics, cell or gene therapies. Sure, health plan sponsors can opt out of step therapy programs but in doing so you expose your company to massive
FWA.