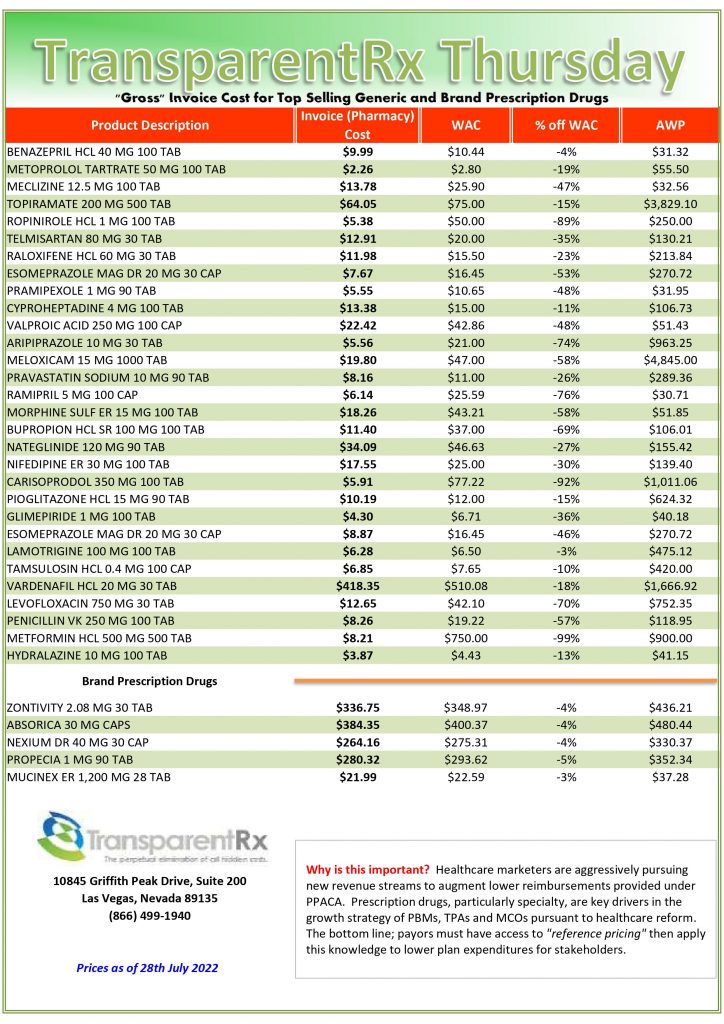
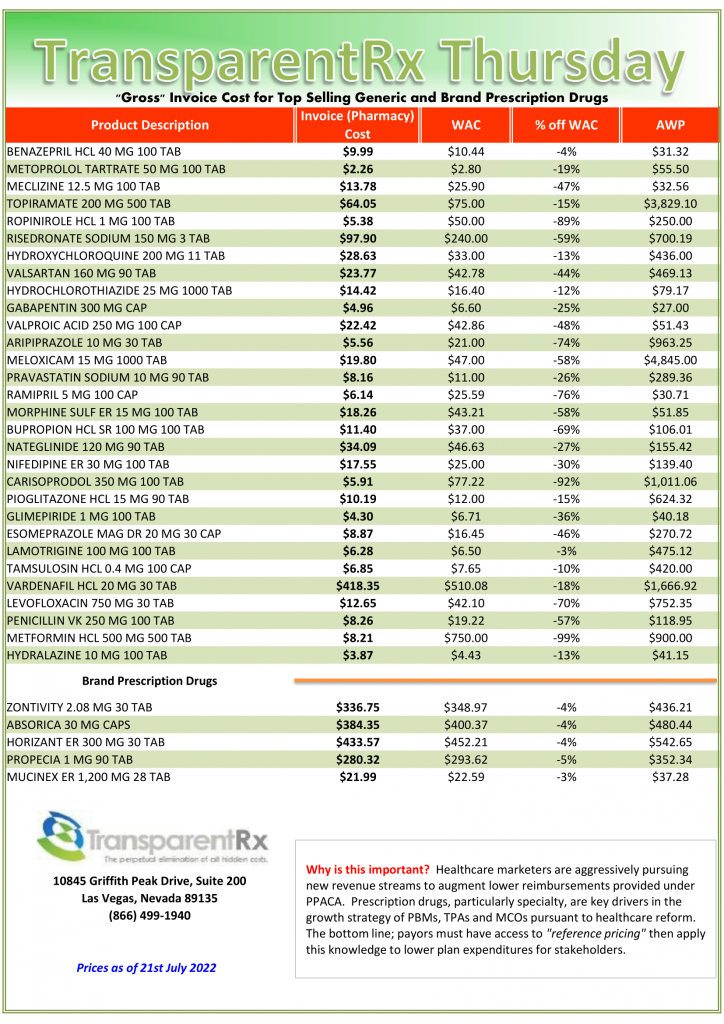
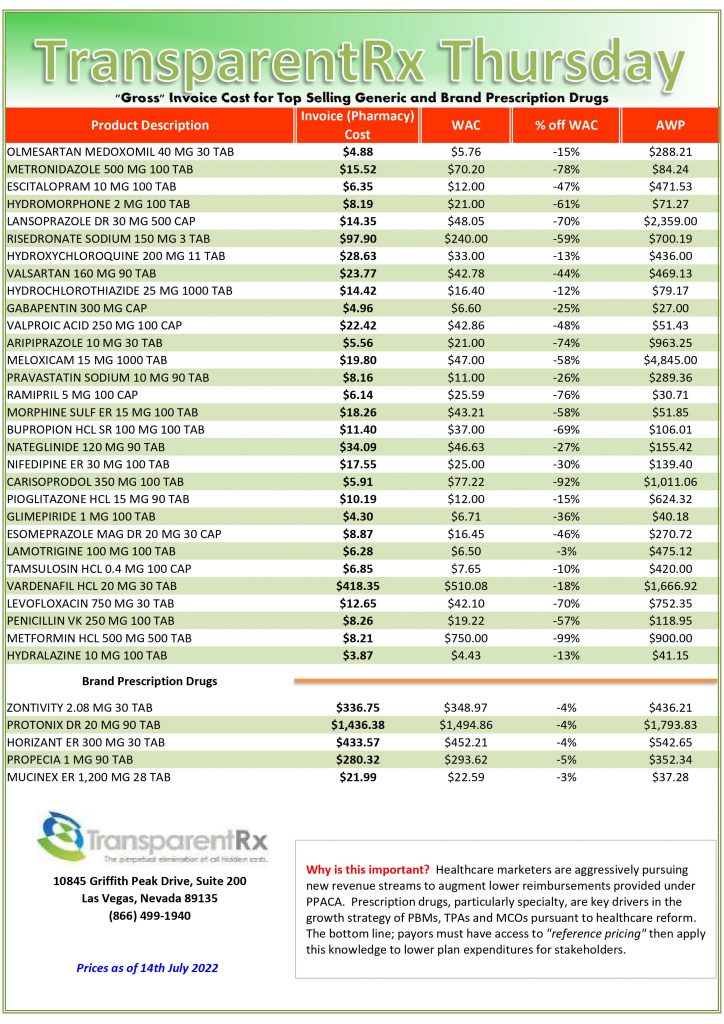
Pharmacogenomics is Increasingly Relevant [Weekly Roundup]
Pharmacogenomics is increasingly relevant and other notes from around the interweb:
- Government declines to appeal ruling in PBM accumulator programs. The Final Rule would have required a manufacturer to count the value of patient cost-sharing assistance in best price and average manufacturer price unless the manufacturer “ensure[d]” that the full value passed to the patient, as opposed to the plan. The Final Rule was vacated on the grounds that it violated the text of the Medicaid rebate statute. We issued a client alert on the ruling available here. The government had 60 days from the date of the ruling to appeal, and that deadline passed on July 18, 2022. Because the government has decided to not appeal the ruling, the Final Rule remains invalidated and will not go into effect.
- Doctors use more brands if they’re on the pharma dole: JAMA. The study offers more evidence that doctors who collect more payments from drugmakers prescribe more brand-name drugs. Harvard Medical School researchers matched Part D Medicare claims in Massachusetts with data on pharma payments to Massachusetts physicians, zeroing in on prescriptions for statin drugs. The widely used class of cholesterol drugs is also widely available in generic form. The conclusion? The higher the payments from drugmakers, the more prescribers used brands, rather than generics. Overall, Massachusetts doctors prescribed branded statins 22.8% of the time. Doctors who collected no money from pharma prescribed brands at a rate of just 17.8%. Doctors who did saw their percentage of brand prescribing rise with every $1,000 in pharma payments reported, the JAMA Internal Medicine study said.
- Precision Medicine vs Trial and Error: Why Pharmacogenomics is Increasingly Relevant. Pharmacogenomics looks at how genetic influences affect an individual’s response to therapeutic medications. Rather than taking the approach that therapy for all people with a certain health condition should begin with the same medication at the same dose, pharmacogenomics has the potential to enable personalized medicine selection and dosing to a degree not previously possible. As of late 2021, 180 approved drugs were labeled by the U.S. Food and Drug Administration (FDA) with genetic factors. While many pharmaco-economic studies suggest that pharmacogenomic testing is cost-effective, a growing body of evidence also highlights the clinical impact of pharmacogenomics. Estimates indicate that more than 98% of people in the U.S. carry at least one high-risk genomic variant in one of the 12 most-tested pharmacogenetic genes. The presence of such variants suggests that treatment with relevant medications may need to be altered to avoid side effects or optimize efficacy. Therefore, the traditional one-size-fits-all, trial-and-error approach to medication management is inherently risky.
- Two firefighters admit role in $50 million prescription fraud scheme. According to the indictment, from July 2014 through April 2016, the conspirators recruited teachers, firefighters, police officers and state troopers enrolled in the state health benefits program to obtain very expensive and medically unnecessary compounded medications, including pain, scar, anti-fungal, and libido creams, as well as vitamin combinations from a Louisiana pharmacy. The pharmacy benefits administrator would pay prescription drug claims and then bill the State of New Jersey for the amounts paid.